Arctic Sea-Ice Controls the Release of Mercury
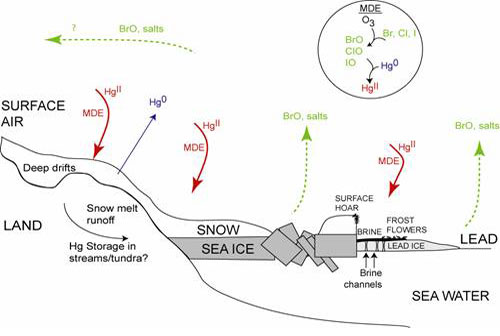
Mercury is the most high profile atmospheric contaminant entering the Arctic because it is a potent neurotoxin that biomagnifies in food webs. In the troposphere (lower atmosphere) it is primarily present in the form of gaseous elemental mercury.  Photochemical reactions during the Arctic spring (Figure 1) combine salts from sea ice and the gaseous mercury in the air to create an oxidized reactive form of mercury. This mercury is then deposited to snow and ice. These deposition events require salty sea ice and snow crystal surfaces so they are widespread in the Polar Regions.
Photochemical reactions during the Arctic spring (Figure 1) combine salts from sea ice and the gaseous mercury in the air to create an oxidized reactive form of mercury. This mercury is then deposited to snow and ice. These deposition events require salty sea ice and snow crystal surfaces so they are widespread in the Polar Regions.
Mercury (Hg) is the only heavy metal that is essentially found in gaseous form in the atmosphere. Since the industrial revolution, emissions of anthropogenic Hg resulting from the combustion of fossil fuels have exceeded natural emissions. Both anthropogenic emissions and natural emissions (which mainly stem from the oceans and gases released by volcanoes) reach the Polar Regions under the action of atmospheric currents. In this way, fallout from global atmospheric pollution contributes to depositing mercury in Arctic ecosystems, even though these are far away from major anthropogenic emission sources.
In the Arctic atmosphere, elementary mercury is oxidized into a form that deposits easily in the cryosphere (snow, ice). Then, when the ice melts, this oxidized form can in turn be re-mobilized and transformed, via physicochemical and biological processes, into a toxin: methylmercury (CH3Hg). It is this toxic form that is ingested by living organisms. It accumulates throughout the food chain and can reach concentrations one million times higher than those measured in surface waters at the very top of the chain. Over the last two decades, mercury and methylmercury concentration monitoring programs in different regions of the Arctic have been showing contrasting geographic and temporal trends. What are the reasons for these variations? What processes govern the mercury cycle?
 To understand these phenomena better, the researchers focused on murre eggs collected in several Arctic and sub-Arctic locations (Gulf of Alaska, Bering Sea and the Chukchi Sea). Situated at the top of the food chain, these sea birds incorporate the mercury contamination present in the chain and are thus an excellent sentinel species for measuring the impact of this pollutant on marine ecosystems. For instance, the quantity of mercury in their eggs provides an accurate reflection of mercury levels in Arctic ecosystems at a given time. More specifically, the team of scientists measured the isotopic signature (2) of Hg in these eggs and noted that it showed significant geographic variations. The isotopic signature variations of most chemical elements (carbon, nitrogen, etc.) mainly depend on their mass difference (12C, 13C).
To understand these phenomena better, the researchers focused on murre eggs collected in several Arctic and sub-Arctic locations (Gulf of Alaska, Bering Sea and the Chukchi Sea). Situated at the top of the food chain, these sea birds incorporate the mercury contamination present in the chain and are thus an excellent sentinel species for measuring the impact of this pollutant on marine ecosystems. For instance, the quantity of mercury in their eggs provides an accurate reflection of mercury levels in Arctic ecosystems at a given time. More specifically, the team of scientists measured the isotopic signature (2) of Hg in these eggs and noted that it showed significant geographic variations. The isotopic signature variations of most chemical elements (carbon, nitrogen, etc.) mainly depend on their mass difference (12C, 13C).
Surprisingly, mercury isotopes do not follow the same “rule”: its odd isotopes (199Hg, 201Hg) behave differently to its even isotopes (198Hg, 200Hg, etc). This particularity is an extremely rare phenomenon on Earth (3). For mercury, this anomaly is closely related to sea-ice cover around murre colonies’ egg laying sites. Knowing the important role played by light in the photodegradation of methylmercury, the researchers succeeded in establishing how much of this toxin could be destroyed by sunlight, whether in the presence or in the absence of sea-ice. In this way, they determined that the presence of sea-ice prevents both the photochemical breakdown of methylmercury and that it limits exchanges of mercury between the Arctic Ocean and the atmosphere.
These results suggest that climate plays a key role in the mercury cycle. Accelerated melting of sea-ice over the coming decades will therefore influence the biogeochemical cycle of this pollutant in a significant manner. Analysis of mercury at the isotopic scale now opens new research avenues to better understand the dynamics of this priority pollutant and its impact on the environment.
This work was initiated within the framework of the 4th International Polar Year (2007-2009) and benefited from ARN support.
(1) Laboratoire des Mécanismes et des Transferts en Géologie (CNRS/IRD/Université Paul Sabatier Toulouse 3/Observatoire Midi-Pyrénées) and the Institut des Sciences Analytiques et de Physico-Chimie pour l’Environnement et les Matériaux (CNRS/Université de Pau).
(2) This indicates the proportions of the different isotopes of a specific chemical element. It can vary both geographically and as a function of time (as is the case with carbon), and can thus indirectly indicate various parameters.
(3) This phenomenon, known as mass-independent isotopic fractionation, was discovered in the 20th century for two light elements in the natural environment: oxygen and sulfur.
-
January 23, 2011 at 3:44 amPoison Dart Frog Pictures |national geographic video | Poison Dart Frog Pictures

![[Most Recent Quotes from www.kitco.com]](https://i0.wp.com/www.kitconet.com/charts/metals/gold/t24_au_en_usoz_2.gif)
![[Most Recent Quotes from www.kitco.com]](https://i0.wp.com/www.kitconet.com/charts/metals/silver/t24_ag_en_usoz_2.gif)

You must be logged in to post a comment.